Support: Creating Reports from Databases/Hosts
Creating from ClinicalTrials.gov
You can import study details from ClinicalTrials.gov using the Bizint Smart Charts Drug Development Suite.
All study fields are imported. Results are not included in the chart at this time.
Important Changes
On June 25, 2024, the "classic" user interface to ClinicalTrials.gov was retired. Version 5.7.4 allows you to build charts from the new generation of ClinicalTrials.gov, following the instructions below.
ClinicalTrials.gov - Step by Step
- Do your search on ClinicalTrials.gov.
- From the List Results window, click the Download button at the top of the results list. The button is outlined in red in the example below.
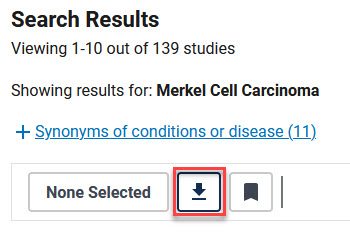
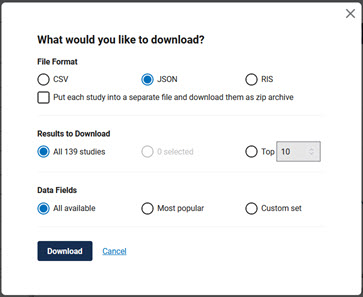
- The "What would you like to download" panel appears. Make the selections as shown below:
- File format: JSON
- You may choose to put each study into a separate file or not - the only difference as far as BizInt Smart Charts is concerned will be the file extension that is downloaded
- Results to download: All NNN studies
- Data fields: All available
- Use File|Import to import the downloaded file (either .ZIP or .JSON depending on which option you choose) into BizInt Smart Charts.
Note: do not unzip the .zip file and import files individually -- import the .zip file itself to import all the documents.



